Subscribe for the latest company news and announcements

Artificial Intelligence (AI)
Singular Health Platform

| Commercialisation Strategy | Pay-per-Scan AI model in 3Dicom; License & Integrate 3rd Party AI Models |
| End-Users | Specialists; Surgeons; Radiologists |
| Main Applications | Triage / Flagging of suspected pathologies; Segmentation of anatomy from CT/MRI scans |
| Competitive Advantage | AI models embedded in single program, run multiple models on one scan simultaneously |
| Outputs | Triage report, highlighting images requiring human review; and, Computed Aided Design files such as STL & OBJ |
Artificial Intelligence Phase – Accelerating Segmentation & Triage Of Medical Images
Since the first FDA-approved AI model in 2014, AI in medicine has grown at a massive 30.4% compound annual growth rate, spawning a USD 1.5 Billion industry.
AI is a complex area, with a large number of approaches and potential applications across medicine from predicting heart attacks and diagnosing disease through to automatically generating drill guides for spinal surgery.
Singular Health views AI as not being essential for the adoption of the Scan to Surgery approach but being an accelerant, reducing the amount of time taken by radiologists to identify and diagnose pathologies (“Triage”) and for surgeons to extract and convert specific anatomical structures into editable surface models (“Segmentation”).
For AI models to be used by end-users, 510(K) regulatory clearance from the FDA and/or TGA regulatory approval is typically required. Singular Health aims to develop internal AI models and seek these clearances in addition to licensing and integrating existing, approved AI models into the 3Dicom software to reduce segmentation workload required for Virtual Surgical Planning.

Singular Health’s AI Projects
Over the past 12 months, Singular Health has developed a number of AI models for various medical applications in collaboration with Australia’s peak science agency, CSIRO, and academics from neighbouring Universities. Many of these models are proofs-of-concept, however Singular Health intends to seek FDA 510(K) clearance for the spinal model developed with CSIRO.
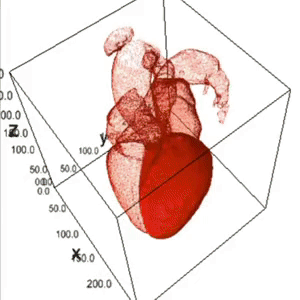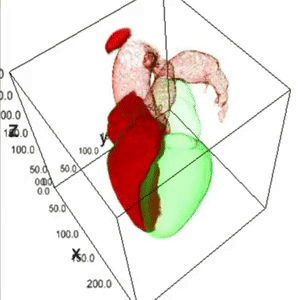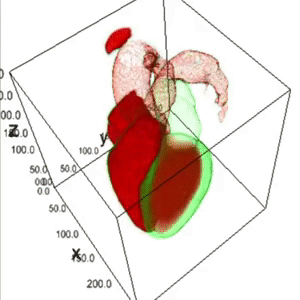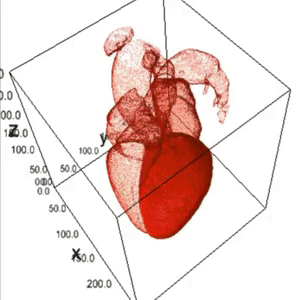
Heart Segmentation

Lung Nodule Detection

Thoracic Segmentation
